Peer Reviewed Articles About Microrganism Found on Floor
Introduction
Human wellness is strongly associated with the balance of microbial communities (Blaser, 2014). The man microbiome provides protection from peel pathogens (Grice and Segre, 2011), aids digestion, supplies nutrients, and activates the immune system (Eckburg et al., 2005; Walia et al., 2014; Adar et al., 2016). A counterbalanced microbial community is more resilient and is improve able to protect confronting pathogen invasion. Dysbiosis impacts the overall stability of a microbial community, leaving the host susceptible to infection and inflammation, with some recent reports showing a link between dysbiosis and immune disorders (Honda and Littman, 2012).
For various macroscale ecosystems, such equally coral reefs, it is well established that greater biodiversity increases the efficiency past which ecological communities can apply essential resources. Coined in 1992, the term holobiont originally divers host-microbe symbioses (Mindell, 1992). Corals found a symbiotic relationship with specific zooxanthellae in their surrounding environments, from which they obtain various nutrients. Corals can also use metabolites from microorganisms and cyanobacteria (Thompson et al., 2014; Cardini et al., 2016). In addition to supplying nutrients, these symbiotic microorganisms are involved in the implantation and development of their host, and provide resistance to pathogens (Thompson et al., 2014).
It is essential to understand holobiotic systems for managing human being health and illness, considering the human microbiome is associated with health outcomes (Postler and Ghosh, 2017). The bacterial limerick is readily altered every bit a upshot of dietary changes, use of antibiotics, infection, and environmental factors (Eckburg et al., 2005; Walia et al., 2014; Adar et al., 2016). In particular, congenital environments, where people spend up to 90% of their time, are likely to influence human health (Klepeis et al., 2001). Each day, the air inhaled by a man typically contains x6 airborne microorganisms (Mandal and Brandl, 2011). Some of these microorganisms crusade pneumonia (e.thou., non-tuberculous mycobacteria (NTM), Legionella and Mycoplasma species), asthma, or allergies (Dannemiller et al., 2016; Montagna et al., 2016; Nishiuchi et al., 2017). More by and large, it has been argued that childhood exposure to reduced levels of microbial diversity in and around homes may partially explicate the ascension in the incidence of allergies and autoimmune disorders in many developed countries (Fujimura et al., 2014; Fang et al., 2016; Human et al., 2017). In high population density indoor environments, such as correctional facilities (Hoge et al., 1994), military machine training centers (Brundage et al., 1988), and dormitories, human being-to-human transmission often occurs. Furthermore, airborne transmission of bacteria in wellness care facilities tin can cause nosocomial infections (Schaal, 1991). The link between mental wellness and the microbiome of the congenital environs is discussed by Hoisington et al. (2015). Therefore, identifying the bacteria present in indoor environments is critically important for human being health.
This review describes the airborne bacteria and its probable sources in built environments (Figures 1, 2). (Many comprehensive reviews of airborne fungi already exist; Prussin and Marr, 2015; Adams et al., 2016 as a result, our review focusses on bacteria).
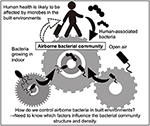
Figure 1. Overview of the present review. Human wellness is probably afflicted by leaner in built environments, because people spend approximately 90% of their lives there. Humans and outdoor air are probable to be the major sources of airborne bacteria as well as bacteria growing in indoors (Burrows et al., 2009; Fahlgren et al., 2010; Bowers et al., 2013).
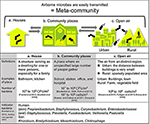
Effigy 2. Built environments were classified into two areas: houses and community spaces. People living and working in such spaces constantly interact with microbes. Airborne microbes also occupy a broad range of built environments. Each blazon of space is continued by air, including open-air environments.
We review the likely factors affecting bacterial concentration and community fluctuations, focusing on temporal variations (temporal and seasonal) and multiscale interactions. The Genomic Standards Consortium (GSC, http://gensc.org) has introduced a system for describing the environment from which a biological sample originates, described every bit "environmental packages." They provided a package for bacterial sequences within built environments (MIxS-Exist (Minimum Information about any (10) Sequence-Built Environment): http://gensc.org/index.php?title=MIxS_extensions) in 2014 (Drinking glass et al., 2014). In this MIxS-BE package, 26 metadata package terms (east.g., carbon dioxide, ventilation type, filter blazon, calorie-free type etc.) are provided too as MIxS-air ecology package terms. These metadata collections could improve indoor microbial community label (Ramos and Stephens, 2014); however, information technology is difficult to collect data on the full set of factors in a typical sampling situation. We therefore, propose four factors (temperature, RH, air commutation rate, and occupant density) that as a minimum should exist routinely measured to monitor and control airborne bacteria in built-upward environments. We also summarize the relevant legislation by land.
Sources of Bacteria
Airborne bacteria can exist treated more effectively if their origin is known. In this section, we summarize the sources of leaner in two chief areas of congenital (house and community places), and open up-air environments (as outlined in Figure 2). Many fungi grow in built environments such as water-damaged homes, schools, and daycare centers, creating severe sanitary issues and potentially being responsible for health problems in humans (Mendell et al., 2011). The impact of airborne fungi on human health means that many state-of-the-art reviews accept been published on this topic (Rao et al., 1996; Dillon et al., 1999; Portnoy and Jara, 2015). In contrast, far fewer reviews have been published on airborne bacteria in congenital environments. Therefore, nosotros have focused on airborne bacteria in built environments in this review.
Bacteria in Houses
There are many sources of bacteria constitute in houses, although the majority of house microbe studies have nerveless samples using surface swabbing equally a proxy for integrated airborne bacterial sampling. Here we draw surface bacteria equally inferred airborne leaner, but for item of the precise sampling method for each study, see Table one "enumeration technique." The principal locations and sources of bacteria in houses are shown in Figure 3. In this review, we hash out the three most mutual sources: human occupants, water, and the outdoor environment, because in our current review, we aim to determine what environmental factors should be measured to understand indoor airborne bacterial communities and concentrations.
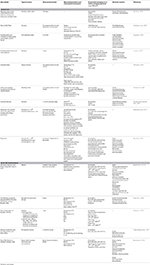
Table i. Changes in microbial customs composition, and the typical sources that influence these changes, from a temporal and seasonal perspective.
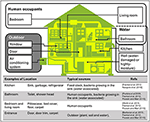
Effigy three. Typical sources of airborne leaner in a house. Three sources are shown: human occupants (white area), water (dotted area), and outdoors (greyness area). Note that these are the groups we take used in this room, but other studies take presented alternatives (Dunn et al., 2013; Prussin and Marr, 2015; Sun et al., 2016).
1 of the sources is human occupants. Virtually of the bacteria institute in houses originate from the peel and abdominal tracts of humans, and include species belonging to the bacterial phyla Proteobacteria, Actinobacteria, Firmicutes, and Micrococcus (Prussin and Marr, 2015; Peccia and Kwan, 2016). When humans motility to a new dwelling, the microbial communities on the surfaces of the new home quickly correlate with those on the pare of the new occupants (Lax et al., 2014). Bacteria from the oral cavity and carrion are predominantly found on pillowcases and toilet seats, respectively (Dunn et al., 2013).
The second major source is water, which disseminates bacteria through the dwelling via showers, taps, and toilets (Perkins et al., 2009; Ichijo et al., 2014). Bacterial growth in buildings may also be facilitated by water leaks, flooring flooding, and in relevant areas, the rainy flavour. Once moisture has accumulated on building surfaces, biocontaminants may proliferate on surfaces then be dispersed as bioaerosols (Fabian et al., 2005). Flooding caused past natural disasters may also promotes mold growth and dispersed of bioaerosols. For example, the tsunami acquired past the Great East Japan Convulsion in 2011 damaged seaside cities. In flood-damaged homes with flooding above flooring level, 63.iii% of respondents (Due north = 256) reported issues after flooding. Mold growth was significantly associated with flooding height (Hasegawa et al., 2015). In the heavily h2o-damaged houses caused by Hurricanes Katrina and Rita in 2005, culturable fungi were significantly higher in moderately/heavily water-damaged houses (geometric mean 76,000 CFU/k3) than in mildly water-damaged houses (geometric mean iii,700 CFU/m3) (Rao et al., 2007).
The 3rd source is the outdoor environs. Airborne bacteria enter houses via natural ventilation components such every bit windows and doors, while other environmental bacteria are transferred from shoes onto floors and carpets (Bouillard et al., 2005). Microbial communities found on floors and carpets probably reflect cultural differences. For example, in Japan, where shoes are removed earlier entering a room, the floor microbial communities are likely to exist dominated by human skin bacteria rather than soil bacteria. Pets and plants are also of import sources of indoor microorganisms. Closed bogus ecosystem experiments in China showed that food stocks in kitchens as well disseminate airborne bacteria (Lord's day et al., 2016).
Information technology should be noted that other researchers accept used culling lists and definitions of these sources for indoor airborne microbes. Sun et al. (2016) monitored airborne microbes in closed artificial ecosystems, and described the possible three sources within that system: (1) human bodies, animals, and plants; (2) microbial contamination during the process of assembling the experimental cabins; and (three) contamination from outside. According to Dunn et al. (2013), they selected nine locations in houses every bit standard sampling locations, based on their existence in virtually all homes: kitchen cutting board, kitchen counter, refrigerator shelf, toilet seat, pillowcase, exterior handle of the main door into the house, television screen, trim of an exterior door, and the upper door trim on an interior door. Prussin and Marr defined eight indoor bioaerosol sources in the built surroundings: humans; pets; plants; plumbing systems; heating, ventilation, and air conditioning systems; mold; resuspension of settled dust; and outdoor air (Prussin and Marr, 2015).
Community Places
Similar to the house microbiome, bacterial communities in offices, classrooms, hospitals and other community spaces are dominated by human and soil bacteria (Hewitt et al., 2012; Hoisington et al., 2016). The master difference between houses and community places is the number of people using the area. People frequenting an surface area can change the microbial community composition of a space, and the number of people and types of human activities are positively correlated with the concentration of bacterial bioaerosols (Heo et al., 2017). To a bottom but significant extent, these factors also influence the composition of indoor bioaerosols (Adams et al., 2015).
Air-mixing is related to many factors: frequency of cleaning, the number of ventilation points, the location of the ventilation organisation, window positions, flooring area, room and building heights, number of occupants, diffuser types, and air speeds and flows. Every bit all of these factors are related to the architectural design of the indoor space, the relationships among ventilation systems, air ducts, frazzle fans, indoor air intake ports, and the microbiome are alluring increasing interest (Kembel et al., 2012; Yang et al., 2016; Zhou et al., 2016). Yang et al. (2016) calculated the fluence rate of the multiple ultraviolet germicidal (UR-UVGI) fixtures system and provide the simulation model, which was validated using the data reported past Rudnick et al. (2015). The results showed that the position of the UR-UVGI fixture almost the outlet accomplished the well-nigh efficient disinfection rate. Of import mechanisms that remove bioaerosols from air include air exchange, deposition onto indoor surfaces, and active filtration. Nazaroff reviewed the dynamic processes that govern indoor concentrations and fats of biological particulate fabric (Nazaroff, 2016). He emphasized that bioaerosol behavior is strongly coupled to particle size (0.1–10 μm). Airborne microorganisms and sampling methods in various selected indoor locations were reviewed by Mandal and Brandl in 2011 (Mandal and Brandl, 2011).
Open-Air Environments
The sources of airborne bacteria are more diverse in open-air environments than in indoor spaces, and include soil, h2o, plants, and insects. The limerick of airborne bacterial communities is influenced by geographical variations such equally landscape and land utilize. For example, ingather harvesting significantly increases the number of airborne bacteria (Elin et al., 1984). In open up-air environments, bacterial sources are difficult to identify because microorganisms are released and transported past diverse organisms and events. For instance, 1 station that was flooded by Hurricane Sandy in the United States all the same resembled a marine surround two years later (Afshinnekoo et al., 2015). Although that report used a swab sampling method rather than an air sampler, such events may also affect airborne microbial communities.
Relationships between Surfaces and Aerosol Loading
Although bacterial sources differ in indoor and outdoor environments, homo-associated and outdoor environmental bacteria dominate the bacterial communities in indoor environments. However, the bulk of house microbe studies nerveless samples by surface swabbing as a proxy for integrated airborne bacterial sampling. The correlation between surfaces and aerosols, and the surface-aerosol interactions in indoor environments, require further investigation. In particular, the book of particles deposited on and and so detached from a item surface is poorly understood. Proving the abovementioned interaction is important for assessing cleaning frequency and tracking the potential impacts of microbes on human health in indoor environments.
Time-Class Analysis of Bacterial Community Limerick
In section Sources of Leaner, we stated that the bulk of bacteria in indoor environments can exist derived from humans, but they depend on many other factors. In some cases, such as low occupant density and high air-modify per hour, human sources are less influential than outdoor bacterial sources (Hospodsky et al., 2015). Therefore, to understand airborne leaner in the built environment, information technology is necessary to clarify both bacterial communities and concentration, also equally environmental factors including human occupation.
Number of Humans Impact the Bacterial Concentration just Not the Communities in Congenital Environments
Humans are surrounded by a vast bacterial ecology (Tyakht et al., 2013; Meadow et al., 2015; Metcalf et al., 2017). These leaner originate from human pare, the oral cavity, and intestine, as well as from clothes. In university classrooms, domiciles, offices, health care facilities and other buildings, the concentration of microorganisms increased during the occupied periods and declined during vacant periods (Stryjakowska-Sekulska et al., 2007; Qian et al., 2012; Park et al., 2013; Hospodsky et al., 2015). According to Qian et al. (2012), all windows and doors were closed during the experiments, and a strong signal of human associated microbes was detected. In the subway systems of Barcelona and Hong Kong, the diversity of airborne microbes was not affected by commuters (Leung et al., 2014; Triado-Margarit et al., 2017). Taxonomic comparison of the microbes from the Hong Kong and New York Metropolis subway networks revealed that Arthrobacter, Psychrobacter, and Enhydrobacter were the predominant bacterial genera in both locations (Robertson et al., 2013). The leaner peradventure originate from the microbial communities of nearby outdoor and human sources. On the Mass Transit Railway (MTR) in Hong Kong, the bacterial diverseness of the collected samples was time-dependent, being more diverse in the afternoons and evening than in the morning. In dissimilarity, the diversity was apparently uninfluenced by commuter traffic (Leung et al., 2014). Airborne microbes around the MTR lines potentially reflect those of nearby outdoor locations, which include soil, water, and leafage-associated organisms (Leung et al., 2014). The point is that in closed-indoor environments, humans are the single chief source of bacteria, so there are different bacterial compositions during occupied and vacant periods. In dissimilarity, in open up-built environments such as MTR, humans are ane of many sources of leaner, and so human-associated bacteria are still present. Varying numbers of humans practice non bear upon bacterial species composition only exercise touch on bacterial concentration. Therefore, humans are probable to affect bacterial concentration simply not influence the overall community variety.
Temporal and Seasonal Variations
Although bacterial communities reportedly vary with the seasons, the factors governing these seasonal variations remain unknown. This is due in part to the multiple bacterial sources in the experimental organization. As described in department Number of Humans Affect the Bacterial Concentration merely Not the Communities in Built Environments, indoor airborne bacterial communities are assumed to be mainly equanimous of man and outdoor environmental bacteria. During indoor air sampling, the microbial fluctuations are mainly correlated with homo occupancy (Table 1). Moreover, they as well depend on occupancy density and levels of human action, such every bit running, walking, sitting, and talking (Qian et al., 2014; Adams et al., 2015; Meadow et al., 2015). All the same, over longer periods, occupants probably utilize natural ventilation (windows and doors), meaning that at least two different bacterial sources are mixed during the experiment (occupants and outdoor). Researchers Hospodsky et al. (2012), Dunn et al. (2013), and Meadow et al. (2014) together combine the fields of microbial ecology, edifice materials and architectural design, to understand microbial variety and affluence within a building. To clarify the individual factors correlated with bacterial community changes, and indoor–outdoor bacterial interaction, the following experiment, originally conducted by Kembel et al. (2012), provides a adept illustration. Researchers collected samples from outdoor air, indoor air from a mechanically ventilated room, and indoor air from a "naturally" ventilated room, simultaneously. The bacterial species related to humans and environments, such as water and soil, were used equally an indicator of indoor bacterial community fluctuation (Dufrêne and Legendre, 1997). They showed that building attributes, specifically the source of ventilation air, airflow rates, relative humidity and temperature, were correlated with the multifariousness and composition of indoor bacterial communities. According to their study design, 2 control experiments are conducted (Figure 4):
(one) Occupants remain, no ventilation during the experiment (e.g., no open windows)
(2) No occupants, windows and doors are open up during the experiment
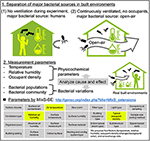
Figure 4. A program for analyzing the causal relationship between seasonal bacterial variations and physicochemical parameters in built environments. The 4 factors (temperature, relative humidity, occupant density, and air commutation charge per unit) are the minimum requirement for monitoring that nosotros recommend.
The physicochemical factors outlined in section Measured Factors at the Time of Airborne Bacterial Sampling in Built Environments are then exist measured in both systems. In organisation (1), the major bacterial source is the occupants. In this instance, human-related bacteria would make upwardly the unabridged bacterial customs in the congenital environment, and we could analyze cause and effect betwixt physicochemical factors and bacterial community composition. A good case of system (1) was studied past Male monarch et al. (2016) in hospitals, where they monitored airborne bacteria in hospital lobbies, waiting areas, corridors, and air ducts, with temperature and humidity maintained at 21°C and forty%, respectively. They institute that the bacterial limerick was relatively stable in hospital environments, suggesting that if in that location is ane bacterial source (such as humans), and environmental factors are stable, the bacterial community would as well exist stable. It is important to note that this discussion concerns bacterial species composition, and non bacterial concentration.
In contrast, in system (2), open-air environmental bacteria would dominate the bacterial community. The problem in this instance is that the effects of ecology conditions on airborne bacterial communities in open-air environments are non well understood, mainly because the bacterial sources are more diverse than in indoor environments. Airborne microbes in open-air environments are likewise influenced past factors such equally wind strength and management, humidity, temperature, and bacterial spore-forming cycles (Fierer et al., 2008). With separating major bacterial sources, analyzing causal relationships between physicochemical parameters and bacterial fluctuations could help u.s. to clarify the factors governing seasonal variation of bacteria. It is interesting to note the factors that were measured when this written report was originally conducted past Kembel et al. (2012): room size and the ventilation rate were measured and compared, assuasive comparison of bacterial communities. Mixed effects models, source-sink assay, and trajectory analysis besides help usa to understand indoor-outdoor bacterial interaction (Frankel et al., 2012; Miletto and Lindow, 2015).
Meta-Communities: Relationships among the Bacteria Inhabiting Indoor and Outdoor Areas
As shown in section Bacteria in Houses, bacterial communities of houses are dominated by human-associated and ecology leaner. In these spaces, the number and composition of bacterial residents are usually stable, and are mainly altered past ventilation (Table two). In contrast, microbial communities in customs spaces are hard to trace, every bit they are dynamically altered by the large amount of foot traffic and the high degree of ventilation. The interaction betwixt indoor and outdoor air has previously been evaluated. The type of ventilation system (artificial or natural), number of ventilation points, and the placement of the ventilation arrangement all affected the concentrations of airborne particles in the indoor and outdoor air samples (Sattar et al., 2016). As seen in section Community Places, many bacteria in community spaces are sourced from occupant behavior, such as opening windows, which also affects the indoor air quality.
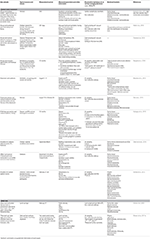
Tabular array 2. Interactions between indoor and outdoor environments, and the influence of outdoor land-use type on other areas.
The relationship between indoor air quality and health has been researched since 1859, when open windows were establish to be essential for maintaining good for you hospital rooms (Nightingale, 1999). Recently, NTM and Legionella infections have go problematic in adult countries. Every bit NTM are ubiquitous in soil and h2o environments, whether infection can exist attributed to the NTM discovered in a patient's dwelling house cannot exist ascertained (Ichijo et al., 2014). Similarly, a report of Legionella infections in healthcare facilities in Italian republic identified the aforementioned Legionella serogroups in air and h2o samples, obscuring the true reservoir (Montagna et al., 2016). To clarify the sources of microorganisms causing infectious diseases, an integrated model is needed (see section Measured Factors at the Fourth dimension of Airborne Bacterial Sampling in Built Environments).
Built environments are non closed systems, and allow the inflow and outflow of many materials. These spaces are also affected by humans and the outside air (Leung and Lee, 2016). Airborne transmission of microbes tin follow different aerodynamic principles. Therefore, we must widen our focus and consider all parameters contributing to the maintenance of a well-balanced microbial customs limerick. Lymperopoulou et al., adopted the term meta-community to describe the microbial composition of the indoor air, which is influenced past microorganisms from environments surrounding the space in question (Lymperopoulou et al., 2016). Therefore, the living space, neighborhood, and whole city can exist regarded as an ecosystem (a meta-community) (Effigy 2).
Measured Factors at the Time of Airborne Bacterial Sampling in Built Environments
We propose measuring four factors (temperature, RH, air substitution rate, and occupant density) at the time of airborne bacterial sampling. Still, this is controversial: some papers country that there is a correlation while others say there is none. In our review, we would like to bear witness that these four factors are the current minimum requirement, since enquiry on airborne leaner in the built environment is ongoing. In studying a microbial community composition, researchers often measure diverse physicochemical values at the time of sampling (Tables i, 2). The GSC provides a package for bacterial sequencing in a built surround (MIxS-BE: http://gensc.org/index.php?title=MIxS_extensions) which includes 26 metadata package terms (Glass et al., 2014; Ramos and Stephens, 2014). The influence of these physicochemical parameters on microbial communities has been investigated in soil and aquatic environments (Stres et al., 2008; Cole et al., 2013). It has also been studied in terms of fungal and bacterial growth in floor dust under elevated and continuous equilibrium relative humidity (ERH). This study indicated that the large increase in microbes at ERH levels >80% and the potent source terms of occupancy-driven resuspension may shape man exposure in buildings under continuous, elevated RH (Dannemiller et al., 2017). The correlation between RH and airborne bacteria in built environments has not been confirmed, although it presumed to influence bacterial growth (Chase et al., 2016). Lax et al. (2017) surveyed the bacterial variety in a newly opened hospital and showed that the bacterial communities nowadays on patients' skin strongly resembled those found in their room surfaces particularly on bedrails. They too analyzed the effect of environmental conditions on microbial transmission and constitute that microbial transmission was correlated with temperature, RH, and humidity ratio. It is non known whether transmission was caused by direct contact or via the air. A model that integrates the physicochemical factors related to transfer and migration, and the concentrations of bacteria in built environments could be useful in some situations (Zargar et al., 2016). In addition, devices that visualize airflow are now available, then the transfer of airborne microorganisms can exist estimated by approximating the air exchange rate and volume of the inside and outside air.
In a infirmary room maintained at constant temperature and RH (25°C and 55%, respectively), fungal and bacterial cells were identified from air samples for at to the lowest degree iii days; nevertheless, past twenty-four hour period 6, no fungal or bacterial colonies were obtained from the air samples (Moungthong et al., 2014). Although these studies cannot ostend whether a sure room temperature and RH suppress the growth of indoor bacteria, they practise suggest that both factors control microbial communities. RH can affect the bacterial customs, while its level of influence is dissimilar in indoor and outdoor spaces. Fierer et al. (2008), for case, establish that indoor RH had no effect on the microbial community composition, simply this composition was significantly correlated with humidity in outdoor air samples. Patient room airborne bacterial sampling conducted by Kembel et al. (2012) showed that edifice attributes, specifically the source of ventilation air, airflow rates, RH and temperature, were correlated with the diversity and composition of indoor bacterial communities. In the MTR samples, outdoor space and, the microbial diverseness and/or abundance of certain genera were influenced by humidity, temperature, CO2 levels, and humans (Robertson et al., 2013; Leung et al., 2014; Afshinnekoo et al., 2015; Triado-Margarit et al., 2017). The influence of ecology factors (temperature, RH, air exchange charge per unit, and occupant density) on bacterial abundance in the air has been reported by some studies, just a full general relationship has not been confirmed. All of these observations support the proposal that microbiome information collection should be accompanied by the measurement of physicochemical factors, such as temperature, humidity, air exchange charge per unit, and occupant density.
Built Ecology Factors and Airborne Microbial Legislation by Country
Humans are constantly exposed to environmental microbes that can touch their microbiome. Here, we reviewed the sources of bacteria and the factors influencing the airborne bacterial communities and concentration in built environments. Bacteria in these spaces originate from different sources, and their communities are directly and indirectly affected by physical factors such as temperature and humidity. To clarify the factors influencing bacterial communities and concentration in homo-occupied spaces, we must standardize the sampling and analysis protocols, equally well every bit the physical parameters (temperature and humidity), as is already done by the Human Microbe Projection (http://hmpdacc.org). Information technology is at present clear that fungal aerosols can cause human disease, and guidelines for fungi in indoor air have existed since 1979 (reviewed past Rao et al., 1996). Moreover, many countries, including the United States, Canada, and France, have established humidity standards for indoor environments because humidity significantly affects the growth of common fungi linked to allergies and breathing problems. China and South korea accept established air quality standards in buildings (Kim et al., 2003; Chan and Yao, 2008), and the American Industrial Hygiene Clan has proposed guidelines outlining the rubber maximum number of fungal spores in different indoor environments (http://www.wondermakers.com). Brazil, Hong Kong, and Singapore accept already regulated the concentrations of airborne microorganisms in indoor environments. Given the health risks posed by airborne microorganisms, which are easily transmitted to dissimilar areas, information technology is important to note that the congenital environs equates to the sum total of all the assembled items that surround u.s., both natural and man-fabricated. By understanding the effects of temperature, RH, air exchange rate, and occupant density on microbial communities in congenital-up areas, we can design healthier living spaces in time to come.
Summary
Ramos and Stephens (2014), Glass et al. (2014), and other researchers in this field depict that there are major issues affecting the study of airborne leaner in built environments, including difficulty in collecting data on the full set of the environmental parameters, and the absence of standardized protocols for data collection, analysis, and interpretation. To empathize the links between airborne leaner and various environmental parameters, as far as possible, the 26 parameters listed by MIxS-BE (http://gensc.org/index.php?title=MIxS_extensions) (encounter Figure 4) should be measured; even so, the sampling situation, materials involved, and diverse other problems, mean that these measurements cannot ever exist taken. Therefore, in line with other researchers, we recommend the routine measurement of four ecology factors (temperature, RH, air substitution rate, and occupant density) to assess airborne bacteria in built environments, every bit a minimum requirement. By improving data collection, we tin begin to understand the airborne bacteria environment of the built surround in more particular every bit a meta-community. This knowledge will provide insights into the relationship between humans and bacterial communities in this environment, and will assist improve our (air) quality of life.
Author Contributions
SF drafted the manuscript, and DT and FM collected and reviewed the literature.
Funding
This work was supported by The Kyoto University Inquiry Funds for Young Scientists to SF, and the Nihon Society for the Promotion of Science KAKENHI (grant number 17K00579, awarded to DT), the Ministry of Teaching, Culture, Sports, Science and Technology of Japan or the Japan Society for the Promotion of Science nether Grants-in-Aid for Scientific Research (KAKENHI) (grant numbers 16H05830/16H05501/16H01782/16H02767, awarded to FM), a Kurita H2o and Environment Foundation Grant, an Ichiro Kanehara Foundation Scholarship Grant for Research in Basic Medical Sciences and Medical Care, a Senri Life Science Foundation Kishimoto Grant, the Nihon Agency for Medical Research and Development (project number 965144, awarded to FM), and by JST/JICA, SATREPS.
Conflict of Interest Statement
The authors declare that the research was conducted in the absenteeism of any commercial or financial relationships that could exist construed every bit a potential conflict of interest.
References
Adams, R. I., Bhangar, S., Dannemiller, Thousand. C., Eisen, J. A., Fierer, N., Gilbert, J. A., et al. (2016). X questions concerning the microbiomes of buildings. Build. Environ. 109(Suppl. C), 224–234. doi: 10.1016/j.buildenv.2016.09.001
CrossRef Full Text | Google Scholar
Adams, R. I., Bhangar, South., Pasut, W., Arens, Eastward. A., Taylor, J. W., Lindow, S. E., et al. (2015). Chamber bioaerosol written report: outdoor air and man occupants as sources of indoor airborne microbes. PLoS ONE ten:e0128022. doi: 10.1371/journal.pone.0128022
PubMed Abstract | CrossRef Full Text | Google Scholar
Adar, S. D., Huffnagle, M. B., and Curtis, J. L. (2016). The respiratory microbiome: an underappreciated player in the human response to inhaled pollutants? Ann. Epidemiol. 26, 355–359. doi: 10.1016/j.annepidem.2016.03.010
PubMed Abstract | CrossRef Full Text | Google Scholar
Afshinnekoo, E., Meydan, C., Chowdhury, Due south., Jaroudi, D., Boyer, C., Bernstein, Due north., et al. (2015). Geospatial resolution of homo and bacterial diversity with city-scale metagenomics. Cell Syst. 1, 72–87. doi: 10.1016/j.cels.2015.01.001
CrossRef Full Text | Google Scholar
Aydogdu, H., Asan, A., and Tatman Otkun, M. (2010). Indoor and outdoor airborne bacteria in child day-care centers in Edirne City (Turkey), seasonal distribution and influence of meteorological factors. Environ. Monit. Assess. 164, 3–66. doi: x.1007/s10661-009-0874-0
PubMed Abstract | CrossRef Full Text | Google Scholar
Barberán, A., Dunn, R. R., Reich, B. J., Pacifici, Yard., Laber, E. B., and Menninger, H. L., et al. (2015). The ecology of microscopic life in household grit. Proc. R. Soc. B 282:20151139. doi: ten.1098/rspb.2015.1139
PubMed Abstract | CrossRef Full Text | Google Scholar
Blaser, M. J. (2014). Missing Microbes: How the Overuse of Antibiotics Is Fueling Our Modern Plagues. New York, NY: Henry Holt and Company.
Bouillard, L., Michel, O., Dramaix, Chiliad., and Devleeschouwer, M. (2005). Bacterial contagion of indoor air, surfaces, and settled dust, and related dust endotoxin concentrations in healthy role buildings. Ann. Agr. Environ. Med. 12, 187–192.
PubMed Abstruse | Google Scholar
Bowers, R. M., Clements, N., Emerson, J. B., Wiedinmyer, C., Hannigan, K. P., and Fierer, N. (2013). Seasonal variability in bacterial and fungal diverseness of the near-surface temper. Environ. Sci. Technol. 47, 12097–12106. doi: 10.1021/es402970s
PubMed Abstract | CrossRef Full Text | Google Scholar
Bowers, R. Chiliad., McLetchie, S., Knight, R., and Fierer, Northward. (2011a). Spatial variability in airborne bacterial communities across land-employ types and their relationship to the bacterial communities of potential source environments. ISME J. 5, 601–612. doi: 10.1038/ismej.2010.167
PubMed Abstruse | CrossRef Total Text | Google Scholar
Bowers, R. One thousand., Sullivan, A. P., Costello, E. G., Collett, J. Fifty. Jr., Knight, R., and Fierer, Northward. (2011b). Sources of bacteria in outdoor air across cities in the midwestern United states. Appl. Environ. Microbiol. 77, 6350–6356. doi: 10.1128/AEM.05498-eleven
PubMed Abstract | CrossRef Full Text | Google Scholar
Bowers, R. M., McCubbin, I. B., Hallar, A. Thou., and Fierer, Northward. (2012). Seasonal variability in airborne bacterial communities at a high-elevation site. Atmos. Environ. 50, 41–49. doi: 10.1016/j.atmosenv.2012.01.005
CrossRef Full Text | Google Scholar
Brundage, J. F., Scott, R. Grand., Lednar, Westward. Grand., Smith, D. W., and Miller, R. N. (1988). Building-associated gamble of febrile acute respiratory diseases in Army trainees. JAMA 259, 2108–2112. doi: ten.1001/jama.1988.03720140028029
PubMed Abstract | CrossRef Full Text | Google Scholar
Burrows, S. Thou., Elbert, Westward., Lawrence, Chiliad. G., and Pöschl, U. (2009). Bacteria in the global atmosphere–Part 1: review and synthesis of literature data for unlike ecosystems. Atmos. Chem. Phys. 9, 9263–9280. doi: 10.5194/acp-nine-9263-2009
CrossRef Full Text | Google Scholar
Cardini, U., van Hoytema, N., Bednarz, Five. N., Rix, L., Foster, R. A., Al-Rshaidat, Thousand., et al. (2016). Microbial dinitrogen fixation in coral holobionts exposed to thermal stress and bleaching. Environ. Microbiol. 18, 2620–2633. doi: 10.1111/1462-2920.13385
PubMed Abstruse | CrossRef Total Text | Google Scholar
Chan, C. G., and Yao, X. (2008). Air pollution in mega cities in Communist china. Atmos. Environ. 42, 1–42. doi: x.1016/j.atmosenv.2007.09.003
CrossRef Total Text | Google Scholar
Chase, J., Fouquier, J., Zare, M., Sonderegger, D. 50., Knight, R., Kelley, S. T., et al. (2016). Geography and location are the primary drivers of part microbiome composition. mSystems 1:e00022-xvi. doi: 10.1128/mSystems.00022-sixteen
PubMed Abstract | CrossRef Full Text | Google Scholar
Cole, J. K., Peacock, J. P., Dodsworth, J. A., Williams, A. J., Thompson, D. B., Dong, H., et al. (2013). Sediment microbial communities in Bang-up Boiling Bound are controlled past temperature and distinct from water communities. ISME J. 7, 718–729. doi: ten.1038/ismej.2012.157
PubMed Abstract | CrossRef Total Text | Google Scholar
Dannemiller, Yard. C., Gent, J. F., Leaderer, B. P., and Peccia, J. (2016). Influence of housing characteristics on bacterial and fungal communities in homes of asthmatic children. Indoor Air 26, 179–192. doi: 10.1111/ina.12205
PubMed Abstract | CrossRef Full Text | Google Scholar
Dannemiller, K. C., Weschler, C. J., and Peccia, J. (2017). Fungal and bacterial growth in flooring dust at elevated relative humidity levels. Indoor Air 27, 354–363. doi: 10.1111/ina.12313
PubMed Abstract | CrossRef Full Text | Google Scholar
Dillon, H. G., Miller, J. D., Sorenson, Westward. One thousand., Douwes, J., and Jacobs, R. R. (1999). Review of methods applicable to the assessment of mold exposure to children. Environ. Wellness Perspect. 107(Suppl. three), 473–480. doi: 10.1289/ehp.99107s3473
PubMed Abstract | CrossRef Full Text | Google Scholar
Dong, L., Qi, J., Shao, C., Zhong, X., Gao, D., Cao, Westward., et al. (2016). Concentration and size distribution of total airborne microbes in hazy and foggy weather. Sci. Total Environ. 541, 1011–1018. doi: 10.1016/j.scitotenv.2015.10.001
PubMed Abstract | CrossRef Full Text | Google Scholar
Dufrêne, M., and Legendre, P. (1997). Species assemblages and indicator species:the demand for a flexible asymmetrical approach. Ecol. Monogr. 67, 345–366. doi: 10.1890/0012-9615(1997)067[0345:SAAIST]2.0.CO;2
CrossRef Full Text | Google Scholar
Dunn, R. R., Fierer, N., Henley, J. B., Leff, J. W., and Menninger, H. 50. (2013). Home life: factors structuring the bacterial diversity constitute within and between homes. PLoS Ane viii:e64133. doi: ten.1371/journal.pone.0064133
PubMed Abstract | CrossRef Full Text | Google Scholar
Eckburg, P. B., Bik, E. M., Bernstein, C. N., Purdom, E., Dethlefsen, L., Sargent, 1000., et al. (2005). Diversity of the human abdominal microbial flora. Scientific discipline 308, 1635–1638. doi: x.1126/science.1110591
PubMed Abstract | CrossRef Full Text | Google Scholar
Elin, R. J., Robertson, E. A., and Sever, Grand. A. (1984). Workload, infinite, and personnel of microbiology laboratories in teaching hospitals. Am. J. Clin. Pathol. 82, 78–84. doi: 10.1093/ajcp/82.one.78
PubMed Abstract | CrossRef Full Text | Google Scholar
Fabian, Yard. P., Miller, S. L., Reponen, T., and Hernandez, M. T. (2005). Ambient bioaerosol indices for indoor air quality assessments of overflowing reclamation. J. Droplets Sci. 36, 763–783. doi: 10.1016/j.jaerosci.2004.11.018
CrossRef Total Text | Google Scholar
Fahlgren, C., Hagström, Å., Nilsson, D., and Zweifel, U. Fifty. (2010). Annual variations in the diversity, viability, and origin of airborne bacteria. Appl. Environ. Microbiol. 76, 3015–3025. doi: 10.1128/AEM.02092-09
PubMed Abstract | CrossRef Full Text | Google Scholar
Fang, Z., Yao, Westward., Lou, Ten., Hao, C., Gong, C., and Ouyang, Z. (2016). Profile and characteristics of culturable airborne bacteria in hangzhou, Southeast of People's republic of china. Droplets. Air Quality Res. sixteen, 1690–1700. doi: 10.4209/aaqr.2014.11.0274
CrossRef Full Text | Google Scholar
Fierer, Northward., Liu, Z., Rodríguez-Hernández, M., Knight, R., Henn, K., and Hernandez, M. T. (2008). Short-term temporal variability in airborne bacterial and fungal populations. Appl. Environ. Microbiol. 74, 200–207. doi: ten.1128/AEM.01467-07
PubMed Abstract | CrossRef Full Text | Google Scholar
Frankel, M., Bekö, G., Timm, One thousand., Gustavsen, S., Hansen, E. W., and Madsen, A. M. (2012). Seasonal variations of indoor microbial exposures and their relation to temperature, relative humidity, and air exchange charge per unit. Appl. Environ. Microbiol. 78, 8289–8297. doi: 10.1128/AEM.02069-12
PubMed Abstract | CrossRef Full Text | Google Scholar
Fujimura, 1000. Eastward., Demoor, T., Rauch, M., Faruqi, A. A., Jang, Due south., Johnson, C. C., et al. (2014). House dust exposure mediates gut microbiome Lactobacillus enrichment and airway immune defense against allergens and virus infection. Proc. Natl. Acad. Sci. U.S.A. 111, 805–810. doi: ten.1073/pnas.1310750111
PubMed Abstruse | CrossRef Total Text | Google Scholar
Glass, E. M., Dribinsky, Y., Yilmaz, P., Levin, H., Van Pelt, R., Wendel, D., et al. (2014). MIxS-Be: a MIxS extension defining a minimum information standard for sequence data from the built environment. ISME J. viii, i–iii. doi: 10.1038/ismej.2013.176
PubMed Abstruse | CrossRef Full Text | Google Scholar
Hasegawa, K., Yoshino, H., Yanagi, U., Azuma, K., Osawa, H., Kagi, N., et al. (2015). Indoor environmental problems and health status in water-damaged homes due to tsunami disaster in Japan. Build. Environ. 93(Pt 1), 24–34. doi: 10.1016/j.buildenv.2015.02.040
CrossRef Full Text | Google Scholar
Heo, K. J., Lim, C. E., Kim, H. B., and Lee, B. U. (2017). Effects of human being activities on concentrations of culturable bioaerosols in indoor air environments. J. Aerosol Sci. 104, 58–65. doi: 10.1016/j.jaerosci.2016.11.008
CrossRef Full Text | Google Scholar
Hewitt, K. M., Gerba, C. P., Maxwell, S. L., and Kelley, Southward. T. (2012). Office infinite bacterial abundance and diverseness in three metropolitan areas. PLoS ONE 7:e37849. doi: 10.1371/journal.pone.0037849
PubMed Abstract | CrossRef Total Text | Google Scholar
Hoge, C. Due west., Reichler, M. R., Dominguez, E. A., Bremer, J. C., Mastro, T. D., Hendricks, 1000. A., et al. (1994). An epidemic of pneumococcal disease in an overcrowded, inadequately ventilated jail. Due north. Engl. J. Med. 331, 643–648. doi: 10.1056/NEJM199409083311004
PubMed Abstract | CrossRef Full Text | Google Scholar
Hoisington, A. J., Brenner, Fifty. A., Kinney, K. A., Postolache, T. T., and Lowry, C. A. (2015). The microbiome of the built environs and mental health. Microbiome 3:lx. doi: 10.1186/s40168-015-0127-0
PubMed Abstruse | CrossRef Full Text | Google Scholar
Hoisington, A., Maestre, J. P., Kinney, 1000. A., and Siegel, J. A. (2016). Characterizing the bacterial communities in retail stores in the United States. Indoor Air 26, 857–868. doi: x.1111/ina.12273
PubMed Abstruse | CrossRef Full Text | Google Scholar
Hospodsky, D., Qian, J., Nazaroff, Due west. W., Yamamoto, N., Bibby, Thousand., Rismani-Yazdi, H., et al. (2012). Homo occupancy as a source of indoor airborne bacteria. PLoS ONE 7:e34867. doi: x.1371/periodical.pone.0034867
PubMed Abstruse | CrossRef Full Text | Google Scholar
Hospodsky, D., Yamamoto, North., Nazaroff, West. Westward., Miller, D., Gorthala, S., and Peccia, J. (2015). Characterizing airborne fungal and bacterial concentrations and emission rates in six occupied children's classrooms. Indoor Air 25, 641–652. doi: 10.1111/ina.12172
PubMed Abstruse | CrossRef Full Text | Google Scholar
Ichijo, T., Izumi, Y., Nakamoto, S., Yamaguchi, North., and Nasu, M. (2014). Distribution and respiratory activity of mycobacteria in household water arrangement of healthy volunteers in Japan. PLoS Ane 9:e110554. doi: ten.1371/periodical.pone.0110554
PubMed Abstruse | CrossRef Total Text | Google Scholar
Kembel, S. W., Jones, Due east., Kline, J., Northcutt, D., Stenson, J., Womack, A. M., et al. (2012). Architectural pattern influences the diversity and structure of the built environs microbiome. ISME J. half dozen, 1469–1479. doi: ten.1038/ismej.2011.211
PubMed Abstract | CrossRef Total Text | Google Scholar
Kim, C. W., Phipps, T. T., and Anselin, L. (2003). Measuring the benefits of air quality improvement: a spatial hedonic arroyo. J. Environ. Econ. Manage. 45, 24–39. doi: 10.1016/S0095-0696(02)00013-X
CrossRef Full Text | Google Scholar
King, P., Pham, 50. Chiliad., Flit, Due south., Sphar, D., Yamamoto, R. T., Conrad, D., et al. (2016). Longitudinal metagenomic assay of hospital air identifies clinically relevant microbes. PLoS ONE 11:e0160124. doi: x.1371/journal.pone.0160124
CrossRef Full Text | Google Scholar
Klepeis, N. Eastward., Nelson, West. C., Ott, Westward. R., Robinson, J. P., Tsang, A. M., and Switzer, P., et al. (2001). The National Human Activity Pattern Survey (NHAPS): a resource for assessing exposure to environmental pollutants. J. Expo. Anal. Environ. Epid. eleven, 231–252. doi: 10.1038/sj.jea.7500165
PubMed Abstract | CrossRef Full Text | Google Scholar
Lax, Southward., Sangwan, North., Smith, D., Larsen, P., Handley, K. G., Richardson, Grand., et al. (2017). Bacterial colonization and succession in a newly opened hospital. Sci. Transl. Med. nine:eaah6500. doi: 10.1126/scitranslmed.aah6500
PubMed Abstract | CrossRef Full Text | Google Scholar
Lax, S., Smith, D. P., Hampton-Marcell, J., Owens, South. M., Handley, M. M., Scott, Due north. Thousand., et al. (2014). Longitudinal analysis of microbial interaction between humans and the indoor environment. Scientific discipline 345, 1048–1052. doi: ten.1126/scientific discipline.1254529
PubMed Abstract | CrossRef Full Text | Google Scholar
Leung, M. H., Wilkins, D., Li, E. 1000., Kong, F. K., and Lee, P. K. (2014). Indoor-air microbiome in an urban subway network: diversity and dynamics. Appl. Environ. Microbiol. lxxx, 6760–6770. doi: x.1128/AEM.02244-fourteen
PubMed Abstract | CrossRef Total Text | Google Scholar
Leung, One thousand. H., and Lee, P. K. (2016). The roles of the outdoors and occupants in contributing to a potential pan-microbiome of the built environs: a review. Microbiome 4:21. doi: x.1186/s40168-016-0165-2
PubMed Abstract | CrossRef Full Text | Google Scholar
Lymperopoulou, D. S., Adams, R. I., and Lindow, Southward. E. (2016). Contribution of vegetation to the microbial composition of nearby outdoor air. Appl. Environ. Microbiol. 82, 3822–3833. doi: 10.1128/AEM.00610-16
PubMed Abstruse | CrossRef Full Text | Google Scholar
Man, W. H., de Steenhuijsen Piters, Due west. A., and Bogaert, D. (2017). The microbiota of the respiratory tract: gatekeeper to respiratory health. Nat. Rev. Microbiol. 15, 259–270. doi: 10.1038/nrmicro.2017.14
PubMed Abstract | CrossRef Full Text | Google Scholar
Mandal, J., and Brandl, H. (2011). Bioaerosols in indoor environment-a review with special reference to residential and occupational locations. Open Environ. Biol. Monit. J. 4, 83–96. doi: 10.2174/1875040001104010083
CrossRef Full Text | Google Scholar
Meadow, J. F., Altrichter, A. E., Bateman, A. C., Stenson, J., Brown, K. Z., Green, J. L., et al. (2015). Humans differ in their personal microbial cloud. PeerJ 3:e1258. doi: 10.7717/peerj.1258
PubMed Abstract | CrossRef Full Text | Google Scholar
Meadow, J. F., Altrichter, A. Due east., Kembel, S. W., Kline, J., Mhuireach, G., Moriyama, M., et al. (2014). Indoor airborne bacterial communities are influenced by ventilation, occupancy, and outdoor air source. Indoor Air 24, 41–48. doi: 10.1111/ina.12047
PubMed Abstract | CrossRef Total Text | Google Scholar
Mendell, 1000. J., Mirer, A. G., Cheung, K., Tong, Yard., and Douwes, J. (2011). Respiratory and allergic wellness furnishings of dampness, mold, and dampness-related agents: a review of the epidemiologic bear witness. Environ. Wellness Perspect. 119, 748–756. doi: 10.1289/ehp.1002410
PubMed Abstruse | CrossRef Total Text | Google Scholar
Metcalf, J. Fifty., Xu, Z. Z., Bouslimani, A., Dorrestein, P., Carter, D. O., and Knight, R. (2017). Microbiome tools for forensic science. Trends Biotechnol. 35, 814–823. doi: 10.1016/j.tibtech.2017.03.006
PubMed Abstract | CrossRef Total Text | Google Scholar
Miletto, 1000., and Lindow, S. E. (2015). Relative and contextual contribution of different sources to the composition and abundance of indoor air bacteria in residences. Microbiome 3:61. doi: 10.1186/s40168-015-0128-z
PubMed Abstract | CrossRef Full Text | Google Scholar
Mindell, D. P. (1992). Phylogenetic consequences of symbioses: eukarya and Eubacteria are not monophyletic taxa. BioSystems. 27, 53–62. doi: 10.1016/0303-2647(92)90046-2
CrossRef Full Text | Google Scholar
Montagna, M. T., Cristina, M. L., De Giglio, O., Spagnolo, A. Thou., Napoli, C., Cannova, L., et al. (2016). Serological and molecular identification of Legionella spp. isolated from water and surrounding air samples in Italian healthcare facilities. Environ. Res. 146, 47–50. doi: x.1016/j.envres.2015.12.015
PubMed Abstract | CrossRef Full Text | Google Scholar
Moungthong, G., Klamkam, P., Mahakit, P., Chalermwatanachai, T., Thunyaharn, Southward., and Monyakul, 5. (2014). Efficacy of the Precise Climate Controller on the reduction of indoor microorganisms. Asia Pac. Allergy four, 113–118. doi: x.5415/apallergy.2014.4.2.113
PubMed Abstract | CrossRef Total Text | Google Scholar
Naegele, A., Reboux, G., Vacheyrou, M., Valot, B., Millon, Fifty., and Roussel, S. (2016). Microbiological consequences of indoor composting. Indoor Air 26, 605–613. doi: 10.1111/ina.12242
PubMed Abstruse | CrossRef Total Text | Google Scholar
Nightingale, F. (1999). Notes on Matters Affecting the Wellness, Efficiency, and Hospital Administration of the British Army Founded Chiefly on the Experience of the Late War, and Notes on Hospitals. Florence Nightingale: Measuring Infirmary Intendance outcomes. Oakbrook Terrace, IL: Joint Commission on Accreditation of Healthcare Organizations.
Nishiuchi, Y., Iwamoto, T., and Maruyama, F. (2017). Infection sources of a mutual non-tuberculous mycobacterial pathogen, Mycobacterium avium complex. Front. Med. 4:27. doi: x.3389/fmed.2017.00027
PubMed Abstract | CrossRef Full Text | Google Scholar
Park, D. U., Yeom, J. K., Lee, West. J., and Lee, K. M. (2013). Assessment of the levels of airborne bacteria, gram-negative bacteria, and fungi in infirmary lobbies. Int. J. Environ. Res. Public Health 10, 541–555. doi: 10.3390/ijerph10020541
PubMed Abstruse | CrossRef Total Text | Google Scholar
Perkins, Due south. D., Mayfield, J., Fraser, V., and Angenent, Fifty. T. (2009). Potentially pathogenic bacteria in shower water and air of a stem cell transplant unit. Appl. Environ. Microb. 75, 5363–5372. doi: 10.1128/AEM.00658-09
PubMed Abstract | CrossRef Full Text | Google Scholar
Postler, T. S., and Ghosh, S. (2017). Understanding the holobiont: how microbial metabolites affect human health and shape the allowed arrangement. Cell Metab. 26, 110–130. doi: 10.1016/j.cmet.2017.05.008
PubMed Abstract | CrossRef Full Text | Google Scholar
Qian, J., Hospodsky, D., Yamamoto, North., Nazaroff, West. W., and Peccia, J. (2012). Size-resolved emission rates of airborne bacteria and fungi in an occupied classroom. Indoor Air 22, 339–351. doi: 10.1111/j.1600-0668.2012.00769.x
PubMed Abstract | CrossRef Full Text | Google Scholar
Qian, J., Peccia, J., and Ferro, A. R. (2014). Walking-induced particle resuspension in indoor environments. Atmos. Environ. 89(Suppl.C), 464–481. doi: 10.1016/j.atmosenv.2014.02.035
CrossRef Full Text | Google Scholar
Ramos, T., and Stephens, B. (2014). Tools to improve built environment data drove for indoor microbial environmental investigations. Buildi. Environ. 81(Suppl. C), 243–257. doi: 10.1016/j.buildenv.2014.07.004
CrossRef Total Text | Google Scholar
Rao, C. Y., Burge, H. A., and Chang, J. C. (1996). Review of quantitative standards and guidelines for fungi in indoor air. J. Air Waste Manag. Assoc. 46, 899–908. doi: x.1080/10473289.1996.10467526
PubMed Abstract | CrossRef Full Text | Google Scholar
Rao, C. Y., Riggs, One thousand. A., Chew, G. L., Muilenberg, 1000. Fifty., Thorne, P. Due south., Van Sickle, D., et al. (2007). Characterization of airborne molds, endotoxins, and glucans in homes in new orleans subsequently hurricanes katrina and rita. Appl. Environ. Microbiol. 73, 1630–1634. doi: 10.1128/AEM.01973-06
PubMed Abstract | CrossRef Total Text | Google Scholar
Rintala, H., Pitkäranta, Thou., Toivola, M., Paulin, Fifty., and Nevalainen, A. (2008). Multifariousness and seasonal dynamics of bacterial community in indoor environment. BMC Microbiol. eight:56. doi: 10.1186/1471-2180-8-56
PubMed Abstract | CrossRef Full Text | Google Scholar
Robertson, C. Eastward., Baumgartner, L. One thousand., Harris, J. K., Peterson, K. L., Stevens, Grand. J., Frank, D. N., et al. (2013). Culture-independent analysis of aerosol microbiology in a metropolitan subway organization. Appl. Environ. Microbiol. 79, 3485–3493. doi: 10.1128/AEM.00331-13
PubMed Abstract | CrossRef Full Text | Google Scholar
Rudnick, S. N., McDevitt, J. J., Hunt, G. Chiliad., Stawnychy, K. T., Vincent, R. L., and Brickner, P. W. (2015). Influence of ceiling fan's speed and direction on efficacy of upperroom, ultraviolet germicidal irradiation: experimental. Build. Environ. 92(Suppl. C), 756–763. doi: 10.1016/j.buildenv.2014.03.025
CrossRef Total Text | Google Scholar
Sattar, Southward. A., Kibbee, R. J., Zargar, B., Wright, One thousand. E., Rubino, J. R., and Ijaz, Chiliad. Thousand. (2016). Decontamination of indoor air to reduce the take chances of airborne infections: studies on survival and inactivation of airborne pathogens using an aerobiology bedroom. Am. J. Infect. Command 44, e177–e182. doi: 10.1016/j.ajic.2016.03.067
PubMed Abstract | CrossRef Full Text | Google Scholar
Schaal, K. P. (1991). Medical and microbiological problems arising from airborne infection in hospitals. J. Hosp. Infect. 18(Suppl. A), 451–459.
PubMed Abstract | Google Scholar
Shin, S. K., Kim, J., Ha, Due south. M., Oh, H. S., Chun, J., Sohn, J., et al. (2015). Metagenomic insights into the bioaerosols in the indoor and outdoor environments of childcare facilities. PLoS ONE x:e0126960. doi: 10.1371/journal.pone.0126960
PubMed Abstruse | CrossRef Full Text | Google Scholar
Smets, W., Wuyts, K., Oerlemans, East., Wuyts, S., Denys, S., Samson, R., et al. (2016). Bear on of urban land employ on the bacterial phyllosphere of ivy (Hedera sp.). Atmos. Environ. 147, 376–383. doi: x.1016/j.atmosenv.2016.10.017
CrossRef Total Text | Google Scholar
Soleimani, Z., Goudarzi, K., Sorooshian, A., Marzouni, M. B., and Maleki, H. (2016). Impact of Heart Eastern dust storms on indoor and outdoor composition of bioaerosol. Atmos. Environ. 138, 135–143. doi: 10.1016/j.atmosenv.2016.05.023
CrossRef Total Text | Google Scholar
Stres, B., Danevcic, T., Pal, Fifty., Fuka, Chiliad. Thou., Resman, Fifty., Leskovec, S., et al. (2008). Influence of temperature and soil water content on bacterial, archaeal and denitrifying microbial communities in drained fen grassland soil microcosms. FEMS Microbiol. Ecol. 66, 110–122. doi: 10.1111/j.1574-6941.2008.00555.ten
PubMed Abstract | CrossRef Full Text | Google Scholar
Stryjakowska-Sekulska, M., Piotraszewska-Pająk, A., Szyszka, A., Nowicki, M., and Filipiak, M. (2007). Microbiological quality of indoor air in university rooms. Smooth J. Environ. Stud. 16, 623–632.
Google Scholar
Lord's day, Y., Xie, B., Wang, Thou., Dong, C., Du, X., Fu, Y., et al. (2016). Microbial customs structure and succession of airborne microbes in airtight artificial ecosystem. Ecol. Eng. 88, 165–176. doi: 10.1016/j.ecoleng.2015.12.013
CrossRef Full Text | Google Scholar
Thompson, J. R., Rivera, H. E., Closek, C. J., and Medina, K. (2014). Microbes in the coral holobiont: partners through evolution, development, and ecological interactions. Front. Cell. Infect. Microbiol. four:176. doi: 10.3389/fcimb.2014.00176
PubMed Abstract | CrossRef Full Text | Google Scholar
Triadó-Margarit, 10., Veillette, M., Duchaine, C., Talbot, Chiliad., Amato, F., Minguillón, Grand. C., et al. (2017). Bioaerosols in the Barcelona subway arrangement. Indoor Air 27, 564–575. doi: x.1111/ina.12343
PubMed Abstruse | CrossRef Full Text | Google Scholar
Tyakht, A. V., Kostryukova, E. S., Popenko, A. South., Belenikin, M. S., Pavlenko, A. V., Larin, A. 1000., et al. (2013). Human gut microbiota customs structures in urban and rural populations in Russian federation. Nat. Commun. four:2469 doi: 10.1038/ncomms3469
PubMed Abstruse | CrossRef Full Text | Google Scholar
Walia, I. S., Borle, R. Thou., Mehendiratta, D., and Yadav, A. O. (2014). Microbiology and antibiotic sensitivity of head and neck infinite infections of odontogenic origin. J. Maxillofac. Oral Surg. xiii, 16–21. doi: 10.1007/s12663-012-0455-6
PubMed Abstract | CrossRef Full Text | Google Scholar
Weikl, F., Tischer, C., Probst, A. J., Heinrich, J., Markevych, I., Jochner, S., et al. (2016). Fungal and bacterial communities in indoor dust follow unlike ecology determinants. PLoS I eleven:e0154131. doi: ten.1371/journal.pone.0154131
PubMed Abstract | CrossRef Full Text | Google Scholar
Yang, Y., Lai, A. C. One thousand., and Wu, C. (2016). Written report on the disinfection efficiency of multiple upper-room ultraviolet germicidal fixtures organization on airborne microorganisms. Build. Environ. 103, 99–110. doi: 10.1016/j.buildenv.2016.04.004
CrossRef Full Text | Google Scholar
Zargar, B., Kashkooli, F. Grand., Soltani, Yard., Wright, K. E., Ijaz, 1000. K., and Sattar, S. A. (2016). Mathematical modeling and simulation of bacterial distribution in an aerobiology sleeping accommodation using computational fluid dynamics. Am. J. Infect. Control 44(9 Suppl.), S127–S137. doi: x.1016/j.ajic.2016.06.005
PubMed Abstract | CrossRef Full Text | Google Scholar
Zhou, P., Yang, Y., Lai, A. C. Chiliad., and Huang, Thousand. (2016). Inactivation of airborne bacteria past common cold plasma in air duct flow. Build. Environ. 106, 120–130. doi: 10.1016/j.buildenv.2016.06.026
CrossRef Full Text | Google Scholar
Source: https://www.frontiersin.org/articles/10.3389/fmicb.2017.02336/full
0 Response to "Peer Reviewed Articles About Microrganism Found on Floor"
Post a Comment